2026 How to Optimize Medical Injection Molding For Global Buyers?
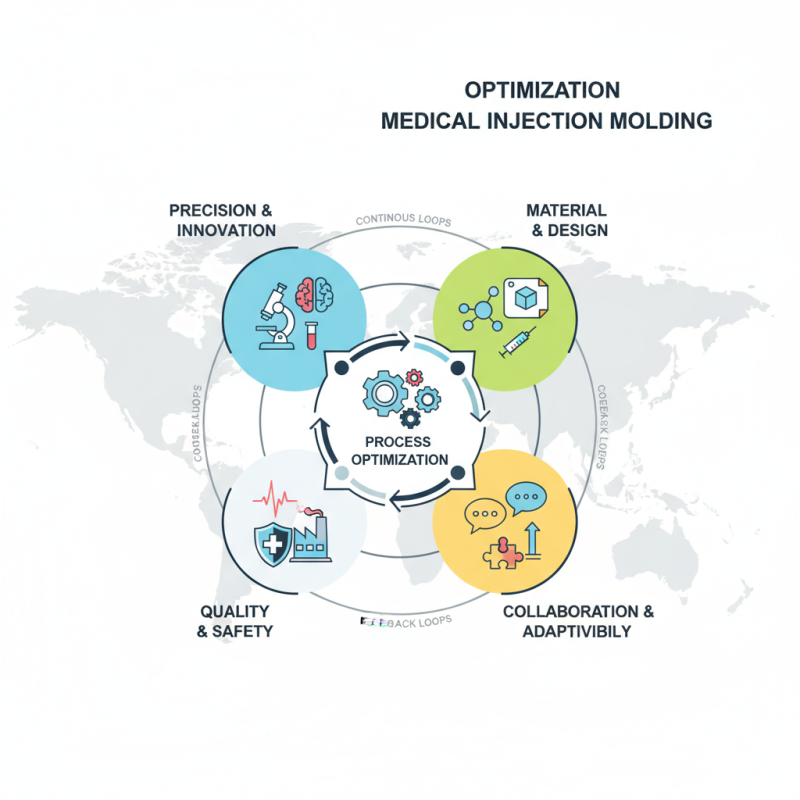
In the rapidly evolving field of medical injection molding, industry experts emphasize the importance of precision and innovation. Dr. Jane Hamilton, a renowned authority in medical device manufacturing, states, "Quality in medical injection molding directly impacts patient safety and device efficacy." This highlights the critical role of optimized processes in ensuring reliable healthcare solutions.
The medical injection molding process serves as the backbone for many essential products. From syringes to complex surgical tools, the accuracy of these components is vital. Engineers must consistently evaluate materials, design, and production methods. However, the industry faces challenges like increasing costs and regulatory demands. Navigating these complexities requires a balance of expertise and practical experience.
Achieving optimal outcomes in medical injection molding isn't just about technology. It demands collaboration across teams. Feedback loops from production to quality assurance are invaluable. There is always room for improvement. Engaging with these challenges can lead to innovative solutions that enhance overall efficiency. The future of medical injection molding hinges on our collective ability to adapt and refine our practices.
Overview of Medical Injection Molding Processes in 2026
In 2026, medical injection molding continues to evolve. The process is crucial for producing precise and sterile medical components. Materials used include thermoplastics like polycarbonate and polystyrene. These materials are favored for their strength and clarity. Each part must meet strict quality standards.
Several key factors influence the molding process. Temperature control is essential for optimal results. Too high or too low can result in defects. The design of the mold itself also impacts product quality. Complex geometries can lead to challenges in manufacturing. It’s important to balance complexity with manufacturability.
Moreover, continuous training for operators is often overlooked. Skilled operators can significantly reduce errors. Regular maintenance of equipment is equally important. Neglecting this can lead to mechanical failures and increased costs. Feedback loops for process improvement are necessary but frequently ignored. Addressing these areas can lead to more efficient production.
Key Considerations for Material Selection in Medical Injection Molding

Selecting the right materials for medical injection molding is crucial. Medical devices require materials that meet specific regulations and safety standards. Factors like biocompatibility and chemical resistance play significant roles. Different applications may require various grades of polymers. Understanding these needs can simplify the selection process.
Thermoplastics often dominate this industry. They offer benefits like durability and ease of processing. However, not all thermoplastics are suitable. Some may degrade when exposed to sterilization processes. It’s essential to have clear communication with suppliers about these properties. Often, manufacturers find themselves needing to revisit material choices midway through production, highlighting a common oversight.
The importance of testing cannot be overstated. Prototype testing reveals how materials will perform under real conditions. Some materials might seem perfect on paper but could fail during application. Regular evaluations ensure that the chosen material truly meets the needs of the final product. Adaptability in material selection is vital for achieving optimal results in medical injection molding.
Advancements in Technology Enhancing Injection Molding Efficiency
Advancements in technology have significantly enhanced the efficiency of medical injection molding. Sophisticated software now allows for precise simulations of the molding process. This helps in predicting potential failures and optimizing designs before physical production. Implementing digital twins enables manufacturers to create a virtual counterpart of the production process. It offers insights that help in minimizing downtime and material waste.
Automation is another critical factor. Automated systems streamline operations by reducing manual intervention. This leads to faster production cycles. However, the reliance on automation raises concerns about the potential for oversights in quality control. Regular monitoring and testing protocols remain essential. Human expertise is invaluable, especially during critical phases of manufacturing.
New materials also play a role in improving molding efficiency. Bio-compatible polymers are increasingly popular. They enhance product safety but require careful handling and testing. Understanding the behavior of these materials during the injection process is still a learning curve for many manufacturers. Investing in research and development is crucial for staying ahead in the industry. Continuous improvements in technology will shape the future of medical injection molding.
2026 How to Optimize Medical Injection Molding For Global Buyers? - Advancements in Technology Enhancing Injection Molding Efficiency
| Dimension | Value |
|---|---|
| Cycle Time | 15-30 seconds |
| Material Types | PP, PET, PVC, Medical Grade Plastics |
| Injection Pressure | 500-3500 psi |
| Dimensional Tolerance | ±0.01 mm |
| Production Volume | 100,000 - 1,000,000 units/month |
| Energy Consumption | Low (< 2 kWh/kg) |
| Automation Level | High (80-90% automated operations) |
| Sustainability Factor | Recycled Materials, Reduced Waste |
Quality Control Measures in Medical Injection Molding for Global Markets
Quality control in medical injection molding is critical for ensuring patient safety and meeting regulatory standards. According to the Medical Device Innovation Consortium, quality issues account for nearly 30% of all product recalls in the medical device sector. This statistic demonstrates the importance of implementing robust quality control measures throughout the production process.
One key measure is the use of statistical process control (SPC). This technique allows manufacturers to monitor processes in real-time, minimizing defects. A report by the International Organization for Standardization indicates that firms employing SPC saw a 25% reduction in defect rates. However, many companies still rely on outdated practices. This reliance can lead to increased variability and product deviations.
Another essential aspect is thorough documentation and traceability. Each step from raw material acquisition to final product testing should be meticulously recorded. Effective documentation helps identify the root causes of issues when they arise. A study by the FDA revealed that lack of proper documentation contributed to 40% of compliance failures in medical device manufacturing. Consequently, businesses must invest in digital tracking systems for enhanced reliability and efficiency. Despite investment requirements, the long-term benefits in compliance and quality make these measures worthwhile.
Sustainability Practices in Medical Injection Molding Techniques
The medical injection molding industry is moving towards greater sustainability. This shift is not just a trend; it's a critical adaptation to increasing environmental concerns. According to a recent report by the Global Industry Analysts, the market for sustainable practices in manufacturing is projected to reach over $50 billion by 2025. Medical injection molding must harness this momentum.
Implementing eco-friendly materials is one primary way to optimize processes. Biodegradable polymers, like PLA and PHA, offer promising alternatives to traditional plastics. These materials can significantly reduce carbon footprints during production. The challenge lies in ensuring they meet stringent medical standards for safety and effectiveness. Not all bioplastics are suitable for medical applications, making material selection crucial.
Another approach is enhancing energy efficiency in production. Studies indicate that energy consumption in injection molding accounts for about 30% of operational costs. Simple changes, like using servo-driven machines, can reduce energy usage by 30% or more. However, the initial investment can be a hurdle for many small manufacturers. Balancing cost and efficiency requires careful planning and evaluation.
2026 Medical Injection Molding Optimization Practices
Related Posts
-

How to Choose the Right Medical Injection Molding Process?
-

Leading Chinese Factory Delivers Unmatched Excellence in Medical Injection Molding
-

Innovative Examples of Automotive Injection Molding Applications in the Industry
-
What is the Impact of Medical Injection Molding on Healthcare Innovation and Patient Outcomes
-

What is Plastic Injection Molding and How Does It Work?
-

How to Understand the Plastic Injection Moulding Process?

